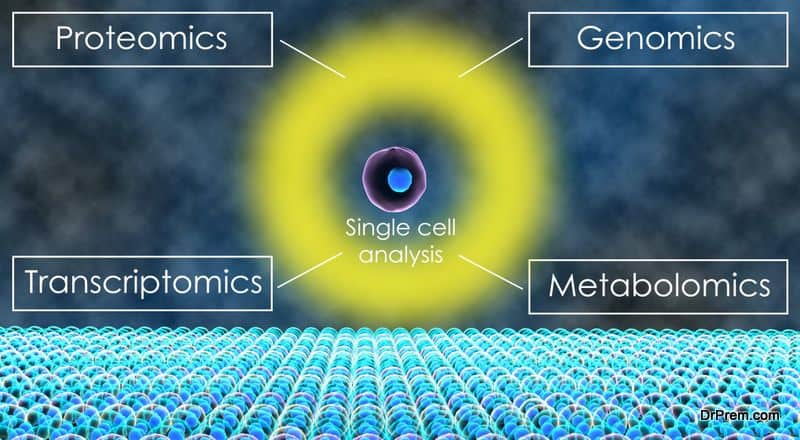
Future global healthcare is aligning to a personalized approach to treatment. Genomics, proteomics, metabolomics and transcriptomics are the emerging terms in medicine supporting this approach. All these come under the OMICS umbrella signifying the study of a whole body of information starting from the cellular level.
Another term that is missing is microbiomics, the study of gut bacteria which might be responsible for various conditions in our body. Microbiomics has been garnering lots of attention in medical research contributing to personalized medicine. The objective is improved treatment outcomes, lesser treatment errors, reduced cost and wastage.
Passing information from one stage to another:
Basically, OMICS involves the study of information processing starting from the DNA until the final expression of genes in RNA passing through various cellular processes. This passage of information is of immense importance as it can provide great insights into disease prediction and personalized treatment which global healthcare is aiming at.
This information processing of genes through different phases is interesting; just like the “Chinese Whisper” game when one message whispered into the beginner’s ear of a chain of participants reaches the final recipient in a distorted form. ‘Chris has a big casket’ can end up in ‘Fish in a basket’.
Systematic analysis of interrelations:
In OMICS, a systematic analysis of interrelations of different pathways of information processing at the cellular level is done taking a holistic approach; thus, the emergence of the vast OMICS universe. It is a deep study of your cellular structure, the RNA, DNA, proteins and metabolites to get a bigger picture of your health status.
How well are you? Are you harboring any ailment whose symptoms are in hibernating mode? Do you have any risk of developing certain chronic ailments in the future? Do you carry any dangerous gene? Can your genes mutate to some life-threatening disease conditions like cancer? All information is recorded in your cellular makeup, OMICS helps to decode it.
OMICs and global healthcare:
OMICS is a relatively new field in medicine but can revolutionize healthcare just by altering the treatment approach. Global healthcare is shifting to data-driven insights, and mining cellular information is important to provide personalized healthcare support which would be more effective, less time consuming and less expensive.
Rapid progress in biotechnology and computing and discoveries from genome sequencing has further opened up opportunities to create precision medicine. Today, mankind can have a look at the innermost secrets of life coded at the cellular level. Decoding this is just a matter of time.
The OMICS Universe:
Let us have an understanding of the different “OMICS” and how they are important in future global healthcare.
Genomics:
The revolution in OMICS in treatment personalization started with genome profiling. It provided valuable insights into elucidating disease mechanisms and molecular diagnoses. The study involves understanding the structure, functioning, evolution, mapping and editing of human genomes or the complete set of DNA.
With the completion of the Human Genome Project, human beings can have a glimpse of the present and future health conditions. Am I susceptible to serious diseases? This is an important question in which genomics is probably giving an accurate answer.
Therefore, the field of medicine that gained substantially from genome sequencing is cancer research. The behavior of different types of cancers, their mutation and genetic expression has been sequenced which is helping in early diagnosis and creating the right treatment plan.
Accomplishments in Genome sequencing so far:
- Discovering the cause of intellectual disability by studying novel genetic mutations in affected candidates
- Treating colorectal cancer
- Rapid whole-genome sequencing of newborns within 50 hours of birth is helping in identifying severe, inherited and treatable genetic diseases.
- Assessing rejection chances of heart transplantation by the recipient
- Cell-free circulating DNA sequencing is used as a biomarker for cancers.
- Development of pharmacogenomics to determine whether a particular medicine, dose or therapy will be effective for a particular patient or not. More than 100 FDA approved drugs carry pharmacogenomics information in their labels.
- An extraordinarily effective drug has been developed for cystic fibrosis by studying gene mutations through genome sequencing
- Whole-genome sequencing has helped in diagnosing and treating rare diseases
Transcriptomics:
The RNA molecules help in the initial transcription of genetic information. A range of RNA molecules functioning in the cell namely, mRNA, small RNAs, non-coding RNAs et al are termed as transcriptome. The mRNA, known as messenger RNA acts as a messenger of passing a part of the DNA code to other parts of the cell for processing.
The Chinese Whisper game starts here. mRNAs are considered vital as they reflect actively expressed genes. Genes express differently owing to exposure to different conditions. Scientists get a new insight into understanding genes and their pathways associated with specific conditions, which would be an indispensable tool in personalized medicine in future.
One application of transcriptomic data is the Oncotype DX Gene Test that analyses the risk of breast cancer recurrence based on RNA transcription. Another application under trial would help in ruling out acute cellular rejection from heart transplants. Transcriptomics also help in understanding the reaction paths to some treatments and infections.
Proteomics:
It is the study of proteins but in OMICS it has a different implication. The second step of cellular information processing takes place with the help of proteins. Proteins receive the transcripted message/information from RNA molecules and process them further. This protein varies from cell to cell and changes over time.
Proteome is a set of information of all proteins expressed in a cell or tissue. It is aimed to understand the functions of proteins in the cells and how the information is passed in the body through different pathways. This functional mechanism of active molecules guides scientists to spot biomarkers which is useful to identify diseases and describe a new class of pharmaceuticals.
Metabolomics:
This is the final product of transcription which implies the study of metabolism and metabolites. Changes in metabolites indicate the shifts in transcriptomes and proteomes which are more complex than the other ‘omes’.
Study of metabolomics help in:
- Understanding various underlying physiological conditions and progression of diseases due to drug action
- Determining environmental and genetic influence on the disease and treatment response
All these contribute a lot to drug development and precision medicine.
Microbiomics:
Talking of future precision medicine, microbiomics is a key component. It involves the study of microbiome or the entire ecosystem of microbes present in our body. The vast dynamic microbial colonies dwelling in our body contribute to a host of metabolic activities to maintain our health.
These colonies also act as trusted guards to prevent a lot of diseases. The growing body of evidence correlating microbiome and early onset of diseases suggests that human microbiome needs to be included in the OMICs universe. This is not the end. We have lipidomic, epigenomics, foodomics and glycomics which might soon open up new scopes of treatment in global healthcare.
The future of OMICs in integrated form:
Increasing research on OMICS since last few years has generated huge data that needs to be processed efficiently to develop precision medicine. The study of individual OMICs field is hugely complex driven by data making it difficult for scientists to get a holistic view. In future, we might achieve something like Unified OMICS test showing the connection between different ‘OMEs’ and diseases.
If such technology can evolve demonstrating the entire lifecycle of a disease starting from genetic mutation and mistranscription of RNA with volumes of proteins ending up in wrong places, and the whole process getting aggravated by some external factors or environment, imagine its impact on future medicine. This will spare physicians from trial and error helping them choose the best medication plan to get maximum efficacy.
AI support in successful OMICs application:
To make OMICs diagnostic fruitful, we need an incredible amount of data for analysis and evaluation which cannot be done without gearing up AI support. OMICs and AI should work together to make precision medicine successful and take preventive and predictive medicine way forward. AI, data analysts and biostatisticians are likely to work with OMICS researchers to successfully guide doctors in OMICS diagnostics for personalized medicine.
Integration of tech and medicine to revamp healthcare based on OMICS:
Technological advancements and medicine research are incredibly innovative providing immense benefits for humanity. But today, there is a need to integrate the organizational cultures of both sectors to bring out the best of them. Global healthcare needs to develop a learning medical system that would collect patient data during providing healthcare and utilize the same to help in treating other patients.
Therefore, data from research and clinical care should not remain in silos. This learning system can be supported by Machine Learning and special software platforms for genomic analysis will not only help in gaining valuable insights but also translate the same in healthcare of a specific population allowing physicians to develop personalized and precision medicine.
Genome sequencing data is growing and so are the challenges:
The first Human Genome Project took 15 years to complete. It is amazing how far we have traveled till date in genome sequencing. Today, it can be done within weeks spending a few hundred dollars. The cost of genome sequencing has dropped by a factor of 1 million in less than a decade!
Researchers assume, 100 million to 2 billion human genome sequencing will be done by 2025. It is overwhelming! Therefore, challenges are growing proportionally regarding data acquisition, storage, distribution and analysis. It needs unique technological solutions. By that time, an estimated 40 exabytes storage may be required for storing genetic information. Some experts believe cloud computing models can help in scaling this data storage requirement.
Apart from this, maintaining data privacy and security is a painstaking issue in global healthcare. Blockchain technology powered platforms are likely to handle this concern effectively facilitating data safety and advanced research in global healthcare through secured data sharing.